|
|
Mechanisms of Viral Entry and Replication
|
|
Project: Lateral Sorting of Influenza Virus
Hemagglutinin in Membranes
Silvia Scolari
Hemagglutinin (HA) is a homotrimeric glycoprotein embedded in the envelope of
Influenza virus. It mediates binding of the virus to the host cell as well as
fusion between the viral envelope and the endosomal membrane. HA is supposed to
entrap in Rafts reflecting liquid-ordered lipid domains enriched of
sphingomyelin and cholesterol.
In order to elucidate the role of the HA transmembrane domain in lipid raft localization we expressed constructs
harboring the transmembrane domain and the cytoplasmic tail but lacking the
N-terminal ectodomain of HA in the plasma membrane of mammalian cells (CHO-K1).
The N-terminus of the transmembrane domain was tagged with YFP (HA-YFP). We
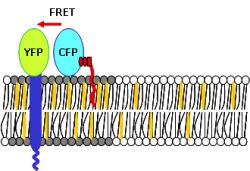
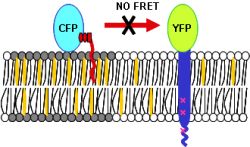
studied Foerster’s energy transfer (FRET) between the artificial HA-YFP and a
GPI anchored CFP as a raft marker by fluorescence lifetime imaging microscopy (FLIM)
(Fig.1). |
 |
First results suggest that HA constructs are indeed sorted and
enriched into cholesterol dependent lipid domains indicated by enhanced FRET
efficiency. This is supported by the observation that cholesterol depletion of
the plasma membrane caused a significant decrease of FRET. Likewise, deletion of
the three highly conserved palmitoylation sites of HA is also accompanied by a
reduction of FRET efficiency. Taken together, the results are in agreement with
sorting of HA constructs into cholesterol-enriched lipid domains.
 |
 |
|
Fig.1: Principle of FRET measurements between yfp-tagged
Influenza HA transmembrane domain and a cfp-bound raft marker. |
References:


