|
|
Mechanisms of Viral Entry, Replication and Assembly
|
|
Project: Lipid-raft association of the HIV glycoprotein
gp41.
A Fluorescence Lifetime Imaging Microscopy study of protein co-clustering in
plasma-membrane microdomains.
Roland Schwarzer
The HIV envelope protein complex mediates host cell infection by binding
cellular receptors and later on triggering membrane fusion. Hitherto, little is
known about the role of those proteins during assembly and budding of the newly
synthesized virus particles. It has been reported that membranes of HIV-infected
cells as well as virions are enriched in cholesterol, hence it was suggested
that HIV-1 may take advantage of cholesterol and sphingolipids during budding.
Both findings point to an important function of lipid microdomains, the so
called rafts, in the late virus lifecycle
 |
|
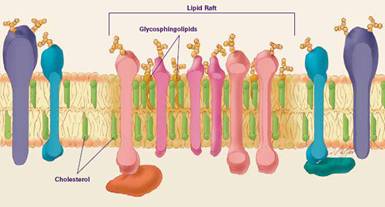
Figure 1: Schematic presentation of suggested plasma membrane
compartmentation
(from website of the National Institute of General
Medical Sciences.) |
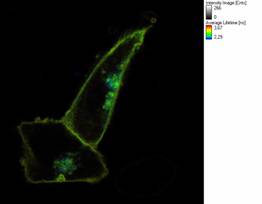
Figure 2: Fluorescence Lifetime (FLIM) image of Chinese Hamster
Ovary cells transfected with fluorescent HIV gp41 and marker
proteins to detect protein clustering and raft localization via FRET. |
This work focuses on the transmembrane part of the viral spike protein
complex formed by the envelope proteins, the glycoprotein gp41. We apply
Fluorescence Lifetime Imaging Microscopy (FLIM) to detect Förster Resonance
Energy Transfer (FRET) between a raft marker, a GPI-anchored cyan fluorescent
protein (CFP), and gp41 fusion proteins labeled with yellow fluorescent proteins
(YFP) to elucidate raft clustering. Since energy transfer is highly dependent on
the distance between the participating molecules, efficient FRET can be
considered as a strong indication for close proximity of raft marker and fusion
proteins and, therefore for colocalization in lipid microdomains. By combining
obtained FRET data with acceptor fluorescence intensity analysis this method
provides reliable clustering information independent of expression level and
fluorophore concentration. Several gp41 chimera were produced to address the
role of different protein domains for raft association but also intracellular
distribution and trafficking. The impact of truncations of the cytoplasmic tail
as well as mutations of, the cholesterol recognition amino acid consensus (CRAC)
domain, intrinsic trafficking signals and a palmitoylation site were studied in
this context.
Results:
Wildtype and several truncated gp41 constructs show FRET upon co-expression with
the raft marker indicating co-clustering in plasma membrane microdomains. The
influence of protein palmitoylation and the CRAC domain on this raft-affinity
will be further investigated.
References:

